FDA Grants Import Discretion of Bracco's Iodinated Contrast Medium Iomeron (iomeprol injection) to Address Supply Shortages
By A Mystery Man Writer
Description
Bracco Diagnostics Inc. has announced that the U.S. Food and Drug Administration (FDA) granted import discretion of Iomeron (iomeprol injection) into the U.S. to address the ongoing iodinated contrast media shortage. The product addresses the need for the most advanced diagnostic imaging standards and will be temporarily available in the U.S. market starting at the end of August, 2022.
articles • APPLIED RADIOLOGY

Contrast Media Imaging Technology News
FDA gives nod to Bracco to import Iomeron to U.S.
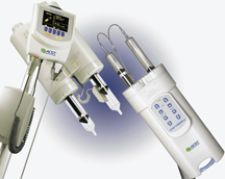
ACIST Features Contrast Injectors, Software

FDA Grants Import Discretion of Bracco's Iodinated Contrast Medium Iomeron (iomeprol injection) to Address Supply Shortages
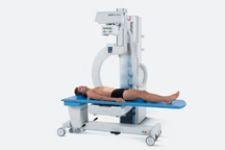
C-Arm System Boasts Technical Innovations
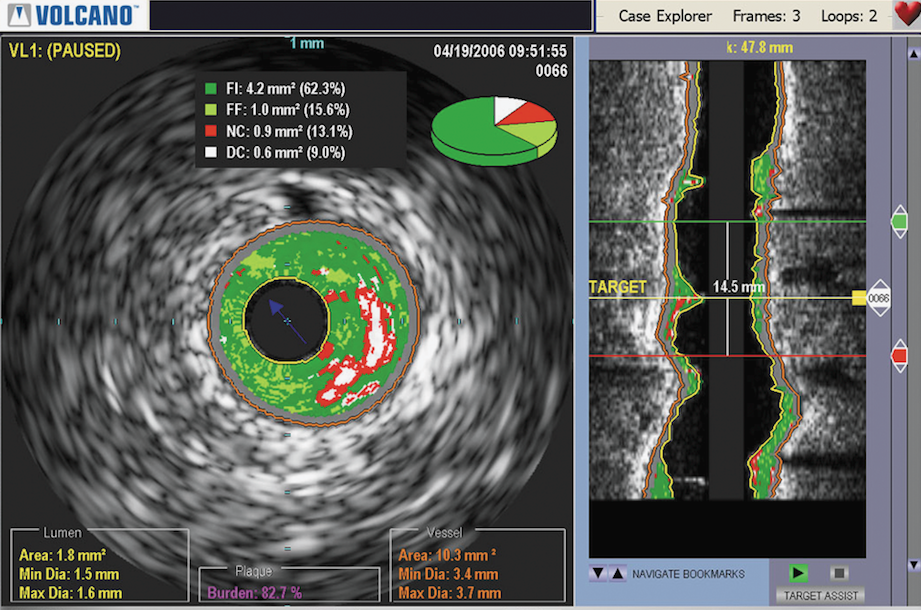
Philips Acquires Volcano for $1 Billion to Expand Position in Interventional Lab Market

Iomeron Injection: Package Insert
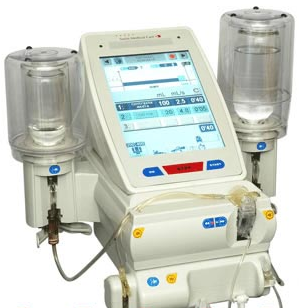
Bracco Acquires Contrast Injector Maker Swiss Medical Care

Definium Family Offers Broad Range of Configurations
Imaging Technology News
from
per adult (price varies by group size)