Acetate [C2H3O2(−)] - Structure, Properties, Molecular Weight & Uses of Acetate ion.
By A Mystery Man Writer
Description
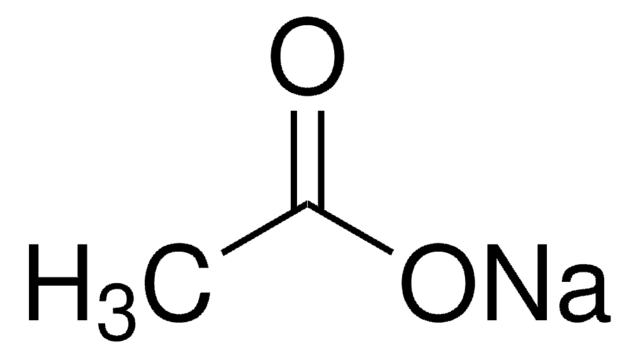
What is an Acetate Ion? - The acetate anion, [CH3COO]−,(or [C2H3O2]−) is one of the carboxylate family. Acetate is a monocarboxylic acid anion resulting from the removal of a proton from the carboxyl group of acetic acid. Visit BYJU’S to learn more about it.
What is an Acetate Ion? - The acetate anion, [CH3COO]−,(or [C2H3O2]−) is one of the carboxylate family. Acetate is a monocarboxylic acid anion resulting from the removal of a proton from the carboxyl group of acetic acid. Visit BYJU’S to learn more about it.
What is an Acetate Ion? - The acetate anion, [CH3COO]−,(or [C2H3O2]−) is one of the carboxylate family. Acetate is a monocarboxylic acid anion resulting from the removal of a proton from the carboxyl group of acetic acid. Visit BYJU’S to learn more about it.
Why is an acetate ion more stable than a phenoxide ion even though the latter is aromatic? - Quora
What is the charge of an acetate ion? - Quora
![Acetate [C2H3O2(−)] - Structure, Properties, Molecular Weight & Uses of Acetate ion.](https://cameo.mfa.org/images/thumb/1/1f/Copper_acetate.jpg/300px-Copper_acetate.jpg)
Copper acetate - CAMEO
![Acetate [C2H3O2(−)] - Structure, Properties, Molecular Weight & Uses of Acetate ion.](https://cdn.clutchprep.com/video_thumbnails/64240.jpg)
Consider the reaction: HC2H3O2(aq) + H2O(l) ⇌ H3O+(aq) + C2H3O2
![Acetate [C2H3O2(−)] - Structure, Properties, Molecular Weight & Uses of Acetate ion.](http://www.t3db.ca/structures/T3D0319/image.png)
T3DB: Lead acetate
![Acetate [C2H3O2(−)] - Structure, Properties, Molecular Weight & Uses of Acetate ion.](https://www.benchchem.com/img/structure/B1219435.png)
Buy Cobalt(II) acetate, 71-48-7
![Acetate [C2H3O2(−)] - Structure, Properties, Molecular Weight & Uses of Acetate ion.](https://www.pw.live/exams/wp-content/uploads/2023/10/All-Shapes-Name-in-English-and-Hindi-73.jpg)
Calcium Acetate Formula, Structure, Properties, Uses
![Acetate [C2H3O2(−)] - Structure, Properties, Molecular Weight & Uses of Acetate ion.](https://pubchem.ncbi.nlm.nih.gov/image/imgsrv.fcgi?cid=22456&t=l)
Lead acetate trihydrate, C4H12O7Pb
![Acetate [C2H3O2(−)] - Structure, Properties, Molecular Weight & Uses of Acetate ion.](https://upload.wikimedia.org/wikipedia/commons/b/b5/Barium_acetate.png)
Barium acetate - Wikipedia
from
per adult (price varies by group size)